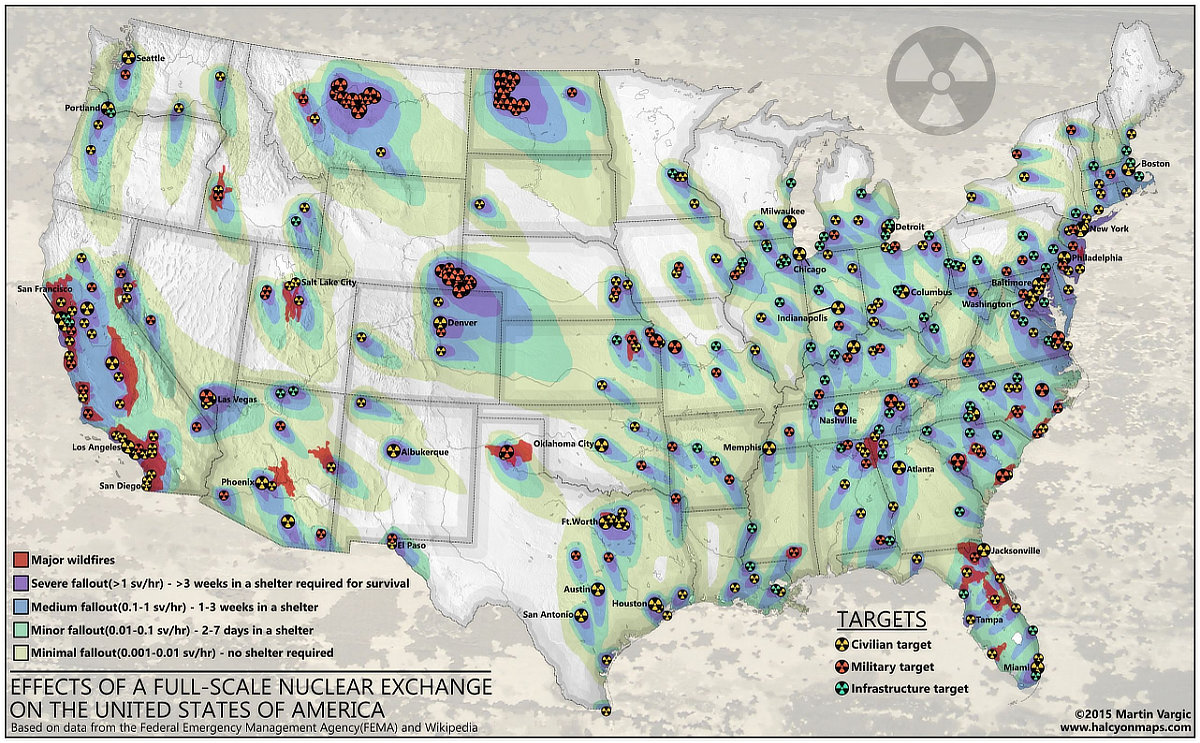
William R. Forstchen is a literary hero of mine going back to grade school. The prolific author and historian has written some of the finest speculative fiction of our time, and in recent years, he has achieved superstardom with the novel ONE SECOND AFTER. It is the story of how a father of two and his small, North Carolina community respond to an electromagnetic pulse (EMP) attack on the United States.
His book has become a touchstone work in the “SHTF” (s— hits the fan) genre, and triggered a national conversation that remains ongoing. An EMP strike would involve a hostile power detonating one or more nuclear weapons over the U.S. at high altitudes. As Congressional testimony described, the explosion would interact with the ionosphere (the electrically conducting layer of the atmosphere) and the Earth’s magnetic field. The resultant surge in electromagnetic currents would essentially fry electronics from coast to coast, cripple the power grid, and cause cascading failures in American infrastructure. The United States is woefully unprepared for such an attack.
Next month, FIVE YEARS AFTER, the fourth book in Forstchen’s exploration of a post-EMP America, hits store shelves. We spoke recently about how an EMP attack would work, what its consequences would be, and how best to prepare for what happens one second after. This interview has been lightly edited for length and clarity.
Where is the EMP threat most likely to come from?
North Korea, followed by Iran. Long ago for nuclear weapons, there was a concept called ‘mutual assured destruction.’ If we launched at the Russians, the Russians would launch at us. Everybody would get blown up and so nobody would win.
But for an EMP, the use of a nuclear weapon is a game changer. It’s known as an ‘asymmetrical first strike.’ All you need is one, two, three small warheads, loft them up over the United States, pop them, and you’ve blinded your enemy. You’ve shut down the power grid. It’s a catastrophic situation.
If that happened, we could find ourselves in a scenario where we don’t even know who the hell launched it. It would be as easy as them using a container ship off the coast of Florida. Launch the weapon, blow the ship up, and where are the fingerprints? Who did it? So this is a very different type of warfare.
What would it take to wipe to fundamentally cripple the United States of America? And how precise do you have to be in detonating these things?
Good question. Really good question. When North Korea launched its first attempts at intercontinental ballistic missiles, I started asking some questions. And people, including from the White House, responded that North Korea doesn’t have EMP capability because ICBMs require a successful launch—it’s got to get up there—and then the warhead has to have a precise guidance system to bring it to its target. Seattle, for instance. It needs a precision reentry for a precision strike.
But you don’t need that with an EMP. All you have to do is launch the weapon up there and get it over the western United States—doesn’t matter at that point if it’s over Seattle, San Francisco, whatever. You don’t need any precision guidance. Therefore they already have the capability.
The ideal scenario is three of them. One in the western United States, one somewhere over the middle—it doesn’t matter if it’s over in Nebraska or Iowa, just pop it—and then one over the eastern United States, say, over Pennsylvania. Pop it. You don’t need precision guidance to do this. It’s like horseshoes: As long as you’re in the general area, you’re going to get some kind of points.
What’s terrifying—and you’ve written great literature on the subject—is what happens next. The real enemy becomes human nature.
Yes, exactly. For a lot of people, you go to the sink and fill a glass of water, and it’s almost miraculous. You turn a faucet and water comes out. If you shut down the electricity, though, you are going to lose your water supply—and that’s true for every major city in the country, because it requires pumping and filtration. What happens to the general populace, within two days, if there is no water? People will be willing to kill to put a bottle of water into their kid’s hand.
Society just starts to break down. The food supply? Twenty days you’re out of food. Medication? The pharmacies are closed. We turn on each other to try and survive, and according to Congressional reporting, upward of 90% of the population will be dead a year later.
One of the strengths the United States has historically enjoyed is geography: we’re just so spread out. How does that play into any sort of post-EMP scenario?
You know, people have this fantasy that if we get hit by an EMP, four or five days into it, you could decide to get the hell out of the city and head to the country. Why? Because there’s a fantasy that out in the country you’ll find food and you’ll find water. Well, even in Nebraska, what do you think they’re dependent upon to bring in the harvest? To plant the seeds? To do all of it? Electricity. But without electricity, all bets are off whether you are in Montana or you are in New York City—it’s all the same.
Does an EMP attack have to be 100% successful to be fundamentally destabilizing? How would we even know we have been hit?
You have to detonate the weapon in the upper atmosphere, the rarefied atmosphere, 200 miles up. The nuclear detonation sets up an electrostatic discharge that then cascades down to the Earth’s surface and blows us out. But it’s not a lightning bolt. You won’t see it, except maybe if you were looking at the point of detonation. Then you’d see a flash, but it wouldn’t even bother your eyesight. You don’t need precision: just put it two hundred miles up, pop it, and it’s a game changer. It’s over.
Tell me about defense policy with respect to retaliation for this sort of thing.
At the end of my book ONE SECOND AFTER, some military people finally come into the town a year later, and one of them says, ‘Oh, yeah, it was the North Koreans who did this to us and we flattened them—we turned their whole country into glass.’ And my main character says, ‘So? What difference does that make now?’
So if this really happened and we retaliated? It would make no difference to any of us because we’d have already lost the war, literally in one second. What we do afterward, it doesn’t matter to you and to me.
So what is Congress doing about this?
I started thinking and writing about the EMP question about 20 years ago, and I based a lot of my book, initially, on Congressional testimony. A number of experts were going up there, all saying the same thing: We’ve got to harden the grid, or we’re waiting to get our butts kicked. There was an attempt at legislative action, but it was killed in committee, particularly thanks to Lisa Murkowski from Alaska, because the bill didn’t have the perks she wanted.
We’ve had at least two or three major efforts to put a bill through. A very strong leader on the issue was Congressman Roscoe Bartlett from Maryland. He was the only person with a PhD in the hard sciences in Congress, and rather than listen to him, they gerrymandered his district to eliminate him. So for 20 years, it’s been gridlocked, and every legislative action has failed.
Now, what are we going to tell those people the day after? Gee, thanks? I had someone say to me more than once that the elite will take care of their own, but the rest of us slobs, we’re doomed. it’s kind of frightening, isn’t it?
So what do you recommend to people who read your book, or who attend your lectures, about what they should be doing in their own lives to prepare for something like this?
To the person who hasn’t done anything: What’s preventing you from getting a month’s worth of supplies in your home? You can do it even if you’re just in a little apartment. Take some two-liter bottles of soda, and when you’re done, clean them out, fill them up with water, and stick them in a closet. You should have at least a couple of gallons of water per day per person in your household.
Next time you go to the market, you don’t need to buy the fancy twenty-five-year shelf life stuff. Just simply pick up Campbell’s Soup and such. That stuff’s good for three or four years. Buy them on sale. Spend fifty bucks a month more than usual to get food, get water.
The third thing I’d say, if you are on medication, don’t wait until the day before the medication comes due to get a refill. If possible, try to keep a three month supply on hand. Next: security. I do not advise people one way or the other regarding guns, but if you decide to do that, I beg you to get trained—and not by your neighbor. Get trained by a professional. I’ve seen too many cases of people getting hurt or killed because of stupidity with guns.
Transportation-wise, you should have a G.O.D. bag in your car: Get Out of Dodge. Just take a backpack, fill it up with two liters of water, a couple of days’ worth of food, one of those space blankets, and keep it in your car. And put some silver in there—actual real silver coins. Because the day after something happens, you could be waving hundred dollar bills and nobody would care. But if you pull out a silver coin, you might be able to trade enough to get what you need. So very basic things like that. They can make a big difference.
There’s this assumption among the public, I think, that if something goes wrong, someone else—likely the government—will take care of everything. But we’ve never encountered a situation where the mechanisms necessary to take care of everything also stop working. Your car stops in the street, and the tow truck that can move it is also stopped. Suddenly, that’s one move too many in a chess game for people to think ahead.
It’s called the expectation of normality. The way things are at this very moment is “normal,” and we expect it will be that way tomorrow and the day after. That’s true for all of us. But if the rug gets pulled out from under us, then what? Suppose on September 10, 2001, a bunch of people started pointing at the sky saying, you know, you could take a couple of jets and turn them into giant cruise missiles. The reaction would be “Oh, you’re crazy. That will never happen.” And then the next day it did.
I keep telling people not to expect normalcy forever. Sooner or later something will happen.
If there’s one consistent, recurring motif in the story of human existence, it is that “something” happens constantly.
Our civilization is temporary. All civilizations are. If you went to the streets of Rome in 150 AD and said, “Guys, it ain’t always gonna be this way. Someday it’s gonna change,” no one would believe you. American civilization, we’ve had 250 years, and we’re doing pretty good. But don’t expect it to run for another 250 years. Something will replace it. Maybe for the better, possibly for the worse. You can’t live on the expectation of normality forever. Every civilization rises, reaches a peak, and collapses. Who’s to say we’re different? We’re not.